H.R. 3, the Elijah E. Cummings Lower Drug Costs Now Act (cited as the Lower Drug Costs Now Act of 2019), awaits a vote by the House in December. The bill has a number of provisions that are intended to make prescription drugs more affordable for those in the Medicare program. A key provision is that it will require the Centers for Medicare and Medicaid Services (CMS) to negotiate prices for some drugs. This is a change from current law, which prohibits the CMS from negotiating the price it pays for drugs. The ability to negotiate prices, combined with the use of the average price paid for that drug in specified countries, along with a top price point of 120% of the reference average list price, is at the heart of the bill.
Under the bill, the maximum price to be paid for any drug will be set through an international comparison of the market price paid for brand name drugs in specified industrialized countries. CMS will negotiate a price that is no more than 120% of that average price. Insulin products are specifically mentioned as among those that CMS must negotiate.
A Painful Pill to Swallow: U.S. vs. International Prescription Drug Prices (the report) prepared by Ways and Means Committee Staff in September 2019 provides the prices used for comparison here. The insulin products included in that report are name brands and will allow us to determine just how much can be saved with a move to an indexed pricing strategy like that proposed in the Lower Drug Costs Now Act of 2019.
Deciding the Average Price
The manufacturer (list) price will be used to calculate the average paid for a drug in six industrialized nations. Currently, those six countries are Australia, Canada, France, Germany, Japan, and the UK. Some refer to these six countries as an Index for the price of a drug. In Europe and elsewhere, drug prices are often set through the use of external reference pricing (ERP).
With ERP, the ex-factory (list) price in a set sample of countries that varies according to the country using that particular ERP is used as a benchmark or basket of prices paid. These prices provide the basis for the negotiation of the price to be paid in the country using the benchmark.
Insulin Pricing in the U.S.
The price paid for insulin products are cited in H.R. 3 as being prices that the CMS must negotiate. The cost of insulin products in the U.S. is currently a matter of great concern. There is widespread news coverage and anecdotal evidence showing that insulin prices are at an all-time high and rising. The report quotes the Centers for Disease Control and Prevention (CDC) as reporting, “… the price of insulin increased by 197 percent from 2002 to 2013. Total Medicare Part D spending on insulin increased by 840 percent from $1.4 billion to $13.3 billion between 2007 and 2017.” A survey of news outlets indicates that, despite differences in the rate price increases of pens versus vials, these trends continue today.
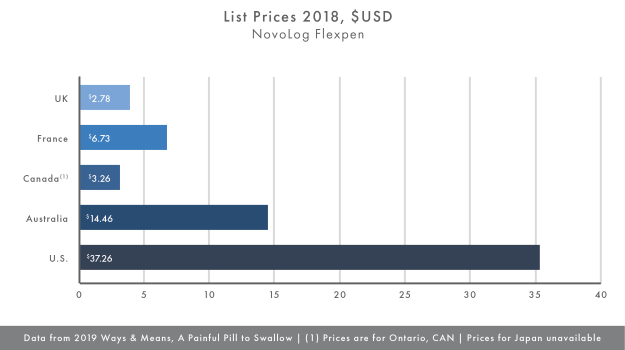
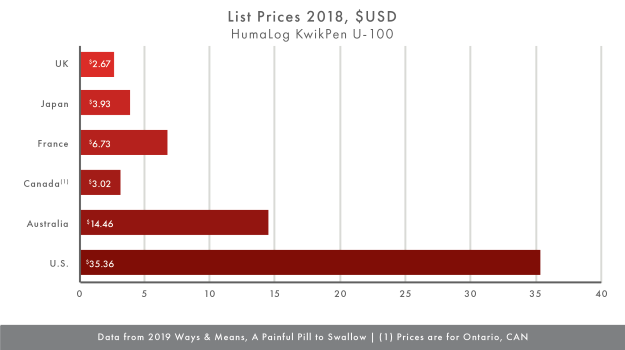
Making use of the data in the report, the average price of Humalog KwikPen U-100 and the NovoLog Flexpen in the reference countries and the U.S. for comparison, Charts I and II show that that the difference in price between the U.S. and other countries is significant. The list price for these insulin products that is closest to the U.S. list price is the price in Australia. With U.S. list prices for the Humalog KwikPen U-100 and the NovoLog Flexpen at $35.36 and $37.26, respectively compared to the Australian list prices of $14.46 for each, the U.S. prices are more than twice as much. When compared to list prices in Ontario, list prices in the U.S. are nearly 12 times as much.
Taking the average list price in the countries in our small sample results in a reference list price for the Humalog KwikPen U-100 of $6.13 and the NovoLog Flexpen of $6.16. Even if CMS used the provision in the H.R. 3 to negotiate a price for these insulin products at 120% of the average, the price in the U.S. would still be six times the average for each product. However, this would represent substantial savings over the current list prices for these products.
What Does it Mean
Clearly, prices in the U.S. exceed those in comparable countries by a wide margin. Even at the slimmest point, list prices in the U.S. are more than twice the other list prices. The claim that pharmaceutical companies report sales their products at a profit in the countries that would be used as reference points, leaves a disturbing disparity in the prices in the U.S. Assuming there are no other variables at work that cause the list prices in the U.S. to carry such a premium while prices in other countries are so much lower, the difference that remains is the vehicle for pricing the drugs. Using reference prices has resulted in lower drug prices around the world. Allowing CMS to negotiate the prices of drugs, within the guidelines of the bill, could effectively make the disparity in list prices for insulin products a thing of the past.
More in our series on H.R. 3, the Elijah E. Cummings Lower Drug Costs Now Act (cited as the Lower Drug Costs Now Act of 2019):
What are the Drugs Most Likely to Remain Undeveloped?
Part IV of our series on the Lower Drug Costs Now Act of 2019 will focus on the drugs least likely to make “the cut” if The Editorial Board of The New York Times is correct in their assessment of economics of the bill.
What are the Alternatives?
In Part V we’ll explore some of the alternatives to the Lower Drug Costs New Act.
The Fate of H.R. 3
The final article in our series will describe the path the bill followed to victory or defeat in the House, including attempts to alter the bill before the vote and the outcome of the vote.
You can also catch up on our series by reading Part I and Part II.
Published on BioSpace
